
Liquid chromatography (LC) is used to separate and analyze the chemical composition of a mixture in solution to determine the presence or absence of a specific component or how much of it is present. High performance liquid chromatography (HPLC) is a column chromatographic separation technique that uses a liquid under high pressure as the mobile phase and a very fine particle size high performance stationary phase.
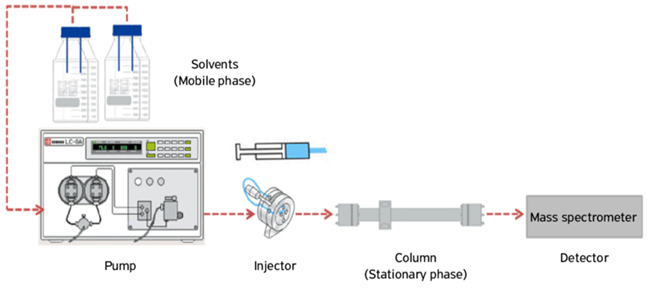 Diagram of the liquid chromatography system (Torre et al., 2015).
Diagram of the liquid chromatography system (Torre et al., 2015).
The autosampler injects the sample into the mobile phase and then carries out the separation process in the column. The selectivity of the chromatographic system has the greatest impact on the chromatographic resolution and should be tailored to the application and components under study. Selectivity can be changed by varying the electrochemical strength of the mobile phase (different solvents) or of the specific chemical functional groups present in the stationary phase (changing the column type).
Considering the mobile phase, there are two main modes of operation available when running a liquid chromatograph, isocratic or gradient. The isocratic method will use the same mobile phase composition for the duration of the chromatographic run with no change in selectivity. The gradient approach will allow the composition of the mobile phase to change over time and can often be optimized to improve chromatographic resolution or reduce run time.
The chemistry of the stationary phase immobilized within the column affects the selectivity of the technique. Reversed-phase HPLC or UHPLC is the most popular system configuration and uses a non-polar stationary phase (e.g., ODS or C18) and a polar mobile phase (water/methanol). Other reversed-phase stationary phases include octasilane (C8), which is less hydrophobic than C18 and has a correspondingly shorter retention time for less polar analytes. If a column functionalized with a phenol substituent is used, this will increase the retention of the phenol fraction due to its increased affinity.
Normal phase chromatography is another liquid chromatography method that separates analytes based on their polarity. In normal phase chromatography the stationary phase is polar while the mobile phase is non-polar. The non-polar components of the mixture are first eluted with the shortest retention time. The polar analytes have a higher affinity for the stationary phase and subsequently elute for a longer retention time. There are other types of liquid chromatography, including ion chromatography, size exclusion, and affinity. For a given set of components to be separated, the selectivity and chromatographic separation obtainable is defined by the stationary and mobile phases used.
Once the analytes are separated, they need to be detected. The choice of detector is driven by the method objectives of the application; various options are available with varying degrees of sensitivity, specificity, selectivity and linear dynamic range. The appropriate detector is selected for subsequent detection based on the requirements.
The development of sub-2 micron silica-based stationary phases with narrow particle size distributions has led to a phase breakthrough in chromatographic performance. The new particles produced have the same chemistry as the commonly used HPLC stationary phases, ensuring that the selectivity of the chromatographic system is maintained while using the same mobile phase. UHPLC offers many advantages, with shorter run times, higher chromatographic separations, higher sensitivity and less solvent consumption.
With UHPLC columns, it is necessary to use pumps that can run at higher pressures to accommodate the increased back pressure exerted by the smaller particles in the column. The detector flow cell also needs to be upgraded to have a smaller internal volume, which is necessary to detect the narrower bands of the components eluting from the column. A corresponding increase in data acquisition rate is also required to ensure sufficient data points between peaks.
Although a wide variety of detectors with different techniques and sensitivities have been coupled to LC for the analysis of different sample types, mass spectrometry has become a selective, sensitive and universal detector.
Unlike other detectors, the LC eluate carrying the separated analytes is not allowed to flow into the mass spectrometer. While the LC system is operating at ambient pressure, the mass spectrometer is operating under vacuum and the two are coupled through an interface. As the column eluate flows into the interface, the analyte molecules are evaporated and ionized by heating to evaporate the solvent. The mass spectrometer can only detect and measure gas-phase ions. Electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) are the most common forms of ionization used in LC-MS analysis.
The analyte ions are drawn into the mass spectrometer. By varying the applied electric and/or magnetic fields to change the flight path of the ions, the ions are separated from each other depending on their charge-to-mass (m/z) ratio. After separation, the ions can be collected and detected by various mass detectors.
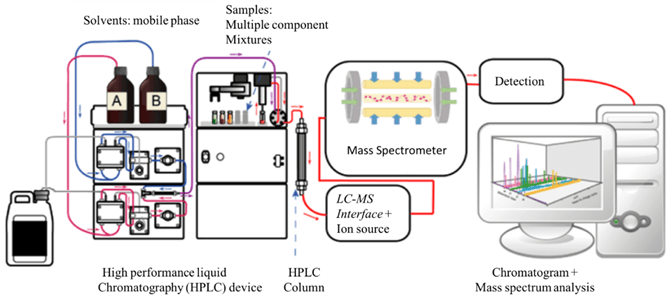
Sample identification and accurate quantification can be further improved by coupling two mass analyzers operating in tandem. Triple quadrupole mass spectrometers (QQQ or TQMS) and quadrupole time-of-flight (QTOF) are the most commonly used tandem mass spectrometers.
Currently, HPLC, UHPLC, LC-MS and LC-MS/MS have been widely used to analyze small molecules in various matrices and can be used to quantify metabolites in biological samples, drug metabolites in biological fluids, genotoxic impurities in active pharmaceutical ingredients, etc.
Creative Proteomics can provide you with metabolomics analysis service based on liquid chromatography and mass spectrometry technology. If you would like more detailed information, please feel free to contact us.
Reference
Copyright © 2026 Creative Proteomics. All rights reserved.