
Plant hormones are highly bioactive signaling molecules that act as chemical messengers, mediating and controlling physiological processes during plant growth and development as well as stress responses by additive, synergistic, or antagonistic actions. Plant hormones are often present in trace amounts and their levels depend strongly on the plant organ, plant developmental stage, and environmental conditions. Plant hormones are structurally diverse active compounds that are divided into several major groups including auxins, cytokinins (CTK), gibberellins (GA), abscisic acid (ABA), ethylene (ETH), jasmonate (JA), salicylic acid (SA), strigolactone (SL) and brassinosteroids (BR). Mass spectrometry is a sensitive analytical technique that enables detection, identification and quantification of a variety of plant hormones by measuring their mass and characterizing their chemical structure, even at low abundance. MS instruments combine with either gas chromatography (GC) or liquid chromatography (LC) to effectively separate organic molecules from complex samples. Comprehensive phytohormone profiling helps elucidate a plant's responses to different conditions such as mutations, stress, and hormone treatments.
Understanding Growth and Development: Analyzing hormone levels helps researchers understand plant growth and development mechanisms.
Environmental Responses: Hormonal signaling reveals how plants adapt to light, temperature, and stress.
Crop Improvement: Manipulating hormone levels enhances crop yield and quality.
Disease and Pest Resistance: Hormone analysis identifies targets for developing disease-resistant crops.
Senescence and Ripening: Ethylene analysis manages post-harvest life of fruits and vegetables.
Root Development and Nutrient Uptake: Studying hormone dynamics in roots informs nutrient uptake strategies.
Environmental Monitoring: Hormones indicate environmental stress on plants.
Biotechnological Applications: Essential for tissue culture and plant transformation.
Ecological Studies: Hormones contribute to plant-plant communication and ecological interactions.
Since different tissues have different matrix properties, Creative Proteomics has developed multiple novel sample preparation techniques. Our services allow the profiling of more than 100 plant hormones, including auxins, GAs, CKs, ABA, BRs, JAs, SA, etc.

Mode: MRM
Precision: pmol
Linear: R2 > 0.99
Analysis content:
| Category | Specific Hormones |
|---|---|
| Auxins | Indole-3-acetic acid (IAA), Indole-3-butyric acid (IBA), 1-Naphthaleneacetic acid (NAA) |
| Abscisic acid | ABA |
| Brassinosteroids | Brassinolide, Castasterone |
| Cytokinins | Zeatin, Isopentenyladenine (IPA), 6-Benzylaminopurine (BAP), Dimethylallyladenine (DZ) |
| Ethylene | Ethylene |
| Gibberellins | GA1, GA3, GA4, GA5, GA6, GA7, GA8, GA9, GA13, GA14, GA15, GA19, GA20, GA24, GA29, GA44, GA51, GA53 |
| Jasmonates | Jasmonic acid (JA), Methyl jasmonate (MeJA) |
| Salicylic acid | Salicylic acid (SA) |
| Strigolactones | Strigolactone |
| Other known hormones | Plant peptide hormones, Polyamines, Nitric oxide, Karrikins, Triacontanol, Phytosulfokine (PSK), Melatonin |
Mass spectrometry-based profiling of plant hormones enables quantitative analyses of plant hormones in a faster, convenient, and sensitive manner. With decades of experience in mass spectrometry services, Creative Proteomics has a proven track record supporting diverse plant hormone detection and quantification. We can meet your specific project requirements, from sampling to bioinformatics.
References
Plant responses to dehydration stress involve complex molecular and hormonal changes. A two-phased response, comprising early stomatal closure and late induction of protective mechanisms, is orchestrated by the hormone abscisic acid (ABA). This study aims to elucidate the temporal dynamics of gene expression and hormone profiles during moderate dehydration stress in Arabidopsis, with a focus on the role of ABA.
Arabidopsis thaliana ecotype Col-0 (WT) and the ABA biosynthetic mutant nced3-2 were employed. Moderate dehydration stress was induced in 3-week-old plants, allowing the separation of early and late stress responses.
Plant Growth and Stress Treatment:
Transcriptome Analysis:
Quantitative Real-Time PCR (qRT-PCR):
Plant Hormone Profiling:
Data Deposition:
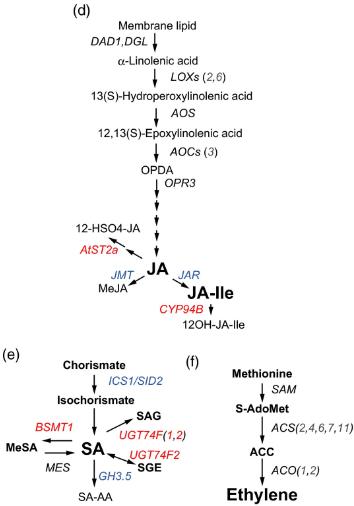 (d) Biosynthetic pathway of (d) JA, (e) SA and (f) ET in Arabidopsis.
(d) Biosynthetic pathway of (d) JA, (e) SA and (f) ET in Arabidopsis.
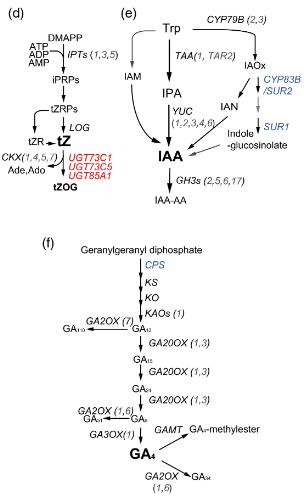 (d–f) Biosynthetic pathway of (d) tZ, (e) IAA, and (f) GA4 in Arabidopsis
(d–f) Biosynthetic pathway of (d) tZ, (e) IAA, and (f) GA4 in Arabidopsis
Reference
Copyright © 2026 Creative Proteomics. All rights reserved.