Gibberellins (GA) Analysis Service
Submit Your InquiryOverview
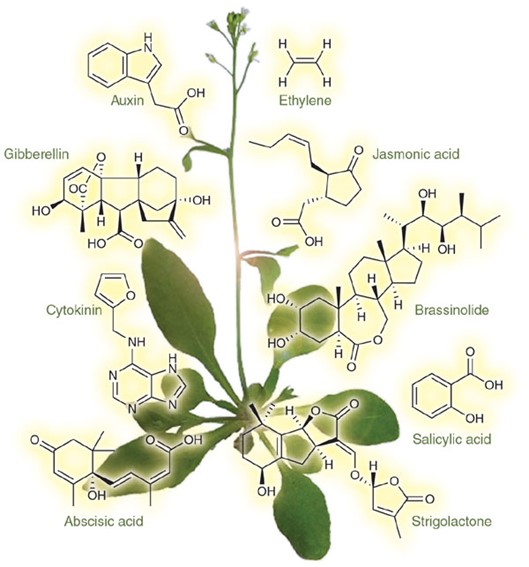
GAs are a class of acid phytohormones present in plants at very low concentrations, normally in the range 10−9–10−15 mol g−1 fresh weight. GAs act as endogenous growth regulators participating in the control of plant growth and development, mainly including stem elongation, germination, flowering and fruit development. Since GAs play essential roles in the developmental processes of plants, it is very important to reveal the GAs functions and the underlying molecular mechanisms. The UPLC method has attracted gradually due to its high sensitivity, short analysis time and significant savings in the cost. The UPLC followed by electrospray ionization-tandem mass spectrometry (ESI-MS/MS) operated in the multiple reaction monitoring (MRM) mode provides a robust and accurate tool for precise quantitation of GAs in various samples in a high selectivity and sensitivity manner. Creative Proteomics offers GA analysis services that have been proved to be a reliable and cost-effective solution for GA detection and quantification in samples with complex matrix.
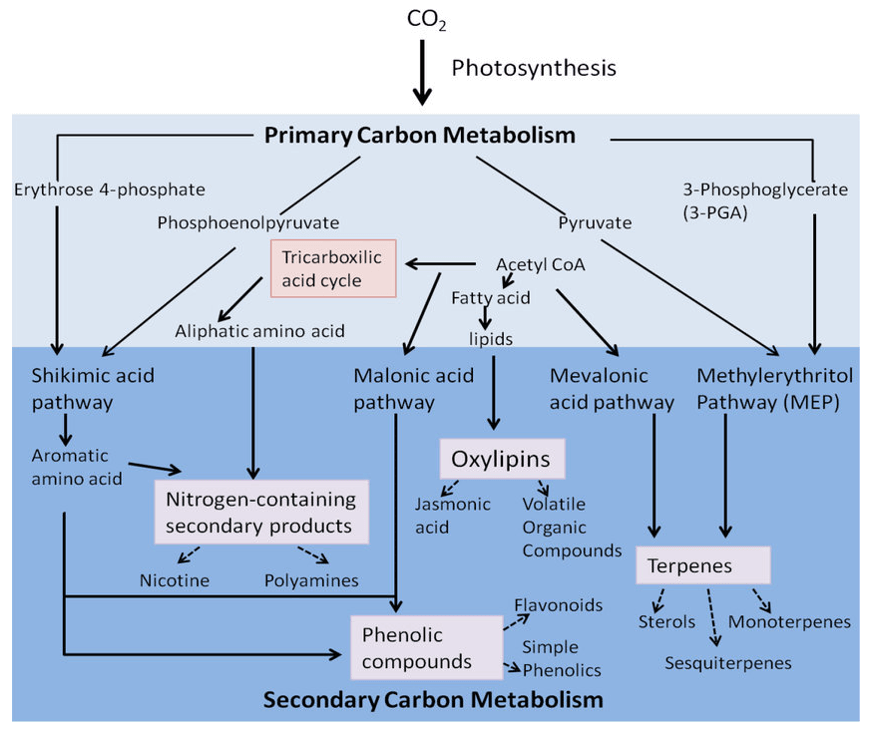
-Analysis-Service-1.png) Figure 1. The gibberellins biosynthesis pathway in higher plants (Igielski & Kępczyńska 2017).
Figure 1. The gibberellins biosynthesis pathway in higher plants (Igielski & Kępczyńska 2017).
Applications of GA Analysis
- Revealing the roles of GAs in plant development
- Elucidating the GA biosynthesis mechanisms and metabolic pathways
- Understanding physiological actions of GAs
- Promoting the application of GAs in agricultural production
Advantages of Our GA Analysis Service
Service Workflow
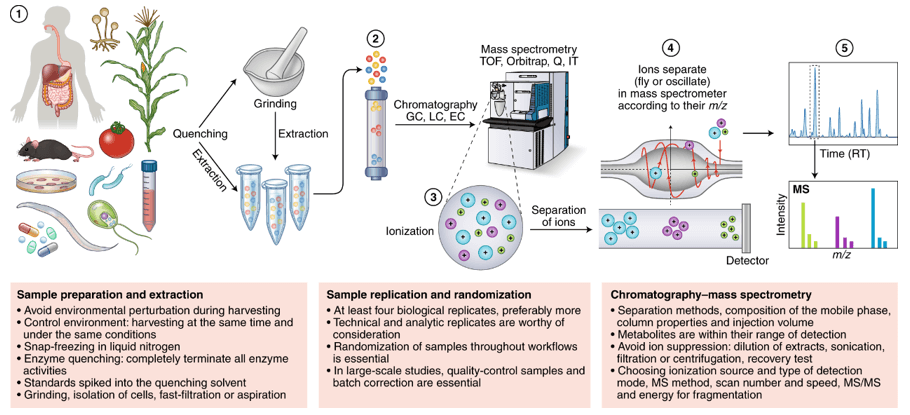
Creative Proteomics provides a highly sensitive and selective method for detecting and quantifying low-concentration GAs in plants using UPLC-ESI-MS/MS. We have strong expertise in handling a variety of sample types, as well as data preprocessing and deep analysis. We ensure the best recovery and ideal GA detection along with reduced background noise.
-Analysis-Service-2.png)
Quantitation method: external standard method / isotope-labeled internal standard method
Analysis content:
- Raw data preprocessing
- Absolute quantitation of GAs
- Differential metabolites screening
- Partial least squares-discriminant analysis (PLS-DA) and principle component analysis (PCA)
List of Detectable GAs at Creative Proteomics
| Gibberellin A1 (GA1) | 545-97-1 | External standard method / isotope-labeled internal standard method |
| Gibberellin A3 (GA3) | 77-06-5 | |
| Gibberellin A4 (GA4) | 468-44-0 | |
| Gibberellin A5 (GA5) | 561-56-8 | |
| Gibberellin A6 (GA6) | 19147-78-5 | |
| Gibberellin A7 (GA7) | 510-75-8 | |
| Gibberellin A8 (GA8) | 7044-72-6 | |
| Gibberellin A9 (GA9) | 427-77-0 | |
| Gibberellin A13 (GA13) | 2922-24-9 | |
| Gibberellin A14 (GA14) | 4955-22-0 | |
| Gibberellin A15 (GA15) | 13744-18-8 | |
| Gibberellin A19 (GA19) | 6980-44-5 | |
| Gibberellin A20 (GA20) | 19143-87-4 | |
| Gibberellin A24 (GA24) | 19427-32-8 | |
| Gibberellin A29 (GA29) | 29774-53-6 | |
| Gibberellin A44 (GA44) | 36434-15-8 | |
| Gibberellin A51 (GA51) | 56978-14-4 | |
| Gibberellin A53 (GA53) | 51576-08-0 |
Sample Requirements
Fresh plant tissue or seed, weight > 1 g. Samples should be frozen in liquid nitrogen right after collection, and then transferred to -80°C for storage.
At least 3 biological replicates.
Delivery
- Experimental procedure
- Parameters of LC-MS
- MS raw data files and MS data quality checks
- GA quantification data
- Custom bioinformatics analysis report
Creative Proteomics's mass spectrometry (MS) facility is equipped with state-of-the-art MS instruments, various ionization techniques (ESI, MALDI, EI, etc.), mass analyzers (Quadrupole, TOFs, Ion Trap and Orbitraps) and separation techniques. We are proud to have been offering MS services for identification, quantitation and mass analysis of plant hormones for years, contributing to reveal their functions, biosynthesis, metabolism, networks, and underlying molecular mechanisms.
References
- Igielski R, Kępczyńska E. Gene expression and metabolite profiling of gibberellin biosynthesis during induction of somatic embryogenesis in Medicago truncatula Gaertn. PLoS One, 2017, 12(7): e0182055.
- Xie W, Han C, Zheng Z, et al. Determination of Gibberellin A3 residue in fruit samples by liquid chromatography–tandem mass spectrometry. Food chemistry, 2011, 127(2): 890-892.
- Urbanová T, Tarkowská D, Novák O, et al. Analysis of gibberellins as free acids by ultra performance liquid chromatography–tandem mass spectrometry. Talanta, 2013, 112: 85-94.
- Hao Y H, Zhang Z, Wang L, et al. Stable isotope labeling assisted liquid chromatography–electrospray tandem mass spectrometry for quantitative analysis of endogenous gibberellins. Talanta, 2015, 144: 341-348.