Service Brochure
Download technical PDF
Accelerate your metabolomics analysis with a full-service solution, combining both untargeted and targeted LC-MS/GC-MS metabolomics.
Untargeted LC-MS/GC-MS for global profiling
Targeted metabolomics panels for absolute quantitation
QC-validated workflows and expert bioinformatics
From small molecules to full pathways, we turn your samples into actionable insights—on time and ready for publication or submission.
Contact ExpertMetabolomics is the large-scale analysis of small-molecule metabolites that reflect the real-time physiological state of biological systems. It enables researchers to connect genotype to phenotype, revealing how cells respond to disease, treatment, or environmental changes.
But modern metabolomics isn't just about generating data—it's about biological clarity, mechanism-focused analysis, and fast, confident decision-making. Whether you're exploring metabolic phenotypes, validating pathway shifts, or integrating multi-omics, what you need is actionable, well-annotated, and publication-ready results—with no guesswork.
That's why our metabolomics services are designed from your research goals backward:
Absolute Quantification with Full Validation
We don't rely on relative intensity. All targeted panels include internal/external standards, 6–8-point calibration curves, and method validation.
Pathway-Level Coverage, Not Just Compound Panels
Our panels are designed around metabolic pathways (e.g., NAD⁺/NADH, methionine cycle, bile acid pool), enabling mechanism-driven analysis.
Untargeted Detection at High Resolution
We routinely detect 1,000–2,000 metabolites per untargeted run with HRMS.
Built-in QC & Batch Correction for Large Studies
Every batch includes pooled QCs, blanks, and internal standards. For >96-sample runs, we provide:
Untargeted metabolomics provides a global, hypothesis-generating view of small molecule metabolites in biological systems. This approach is ideal for:
We detect and relatively quantify hundreds to thousands of metabolites across sample types. Suitable for early-stage research, comparative studies, and systems biology applications.
Targeted metabolomics focuses on quantifying a predefined set of metabolites with high accuracy and sensitivity. It is hypothesis-driven, often used for:
Our targeted metabolomics panels cover a wide range of compound classes and are customizable for human, plant, and microbial studies. All measurements are based on internal/external standards with LOD/LOQ and QC validation.
| Aspect | Untargeted Metabolomics | Targeted Metabolomics |
| Purpose | Explore unknown metabolites and generate hypotheses | Precisely quantify known metabolites |
| Approach | Broad, unbiased screening | Focused, hypothesis-driven analysis |
| Analyte Scope | Hundreds to thousands of features | Dozens to hundreds of predefined compounds |
| Quantification | Relative quantitation | Absolute quantitation (internal standards, calibration curves) |
| Sensitivity | Moderate | High |
| Typical Use Cases | Early discovery, pathway screening, phenotype comparison | Biomarker validation, drug response, mechanistic studies |
| Flexibility | High – suitable for unknowns | Fixed or customizable panels |
| Data Output | Feature table, MS/MS annotations, PCA, volcano plot, pathway enrichment | Concentration tables, LOD/LOQ, CV%, validation metrics |
| Recommended When... | You don't know what to expect and want full coverage | You have clear targets and need robust quantitation |
Our bioinformatics team supports pathway-level interpretation through advanced statistical modeling and visual analysis. Services include:
Understand not only what changes, but how fast metabolites flow through biological pathways. Creative Proteomics offers 13C-labeled metabolic flux analysis, enabling:
Available as an add-on for microbial, plant, or mammalian cell systems. Contact us to discuss tracer substrates, modeling approaches, and data integration options.
| Category | Examples |
| Central Carbon Metabolism | Central carbon metabolism, Glycogenesis, Gluconeogenesis |
| Amino Acid Metabolism | Urea cycle metabolism, Methionine cycle metabolism, One-carbon metabolism, BCKA metabolism |
| Energy Metabolism | Energy metabolism, NAD⁺/NADH balance, β-oxidation flux |
| Lipid Metabolism | Lipid metabolism, Acylcarnitine metabolism, Oxylipin metabolism |
| Carbohydrate Metabolism | Carbohydrate metabolism, Glycan metabolism |
| Nucleotide Metabolism | Nucleotide metabolism (2) |
| Plant-Specific Pathways | Plant hormone pathways, Secondary metabolite pathways |
| Redox & Stress Response | ROS metabolism, Redox cofactors, Glutathione metabolism, Oxidative stress markers |
| Environmental & Toxicology | Xenobiotic metabolism, Glyphosate pathway |
| Other Specialized Pathways | Bile acid metabolism, Ethanol metabolism, Urolithin A metabolism, TMAO metabolism |
At Creative Proteomics, we utilize a full suite of LC-MS and GC-MS platforms, enabling high-resolution, high-throughput metabolomics across diverse sample types. Our systems support both untargeted discovery and targeted quantitation workflows with precision, reproducibility, and wide chemical class coverage.
Our platforms are optimized for:
Key Technical Parameters
| Metric | Typical Capability |
| Mass Accuracy | ≤ 5 ppm (HRMS) |
| LOD/LOQ | < 1 nM (compound-dependent) |
| Dynamic Range | 104–106 |
| Reproducibility (QC CV%) | < 15% |
| Ionization Modes | Positive & Negative (ESI/APCI) |
| Quantitation Types | Absolute (w/ standards) or Relative |

Thermo Q ExactiveTM series

AB Sciex 6500+

Thermo Orbitrap Fusion Lumos

Waters Xevo TQ-s

Thermo TRACE 1310-ISQ LT

Thermo TSQ 9000

Agilent 6495 Triple Quadrupole LC/MS Coupled with the Agilent 1290 Infinity II LC System

ACQUITY UPLC
Sample Preparation & QC
Handles diverse sample types with standardized extraction. Internal standards and QC pools ensure stability and normalization.
Instrumental Analysis (LC-MS / GC-MS)
UPLC or GC separates metabolites; HRMS enables untargeted profiling, triple quadrupole MS supports targeted quantification. Both ion modes broaden coverage.
Data Acquisition & Processing
Collects full-scan MS/MS spectra. Data undergo peak detection, alignment, and QC-based normalization.
Identification & Quantification
Matches spectra to HMDB, KEGG, METLIN, or in-house libraries. Provides relative (untargeted) or absolute (targeted) quantification with functional annotation.
Statistical & Bioinformatics Analysis
Applies PCA, PLS-DA, volcano plots, and heatmaps to detect significant changes. Maps metabolites to KEGG, Reactome, and HMDB pathways.
Pathway & Biological Insights
Performs enrichment and network analysis; optional 13C MFA reveals flux dynamics. Highlights key pathways, biomarkers, and mechanisms.
Please refer to the table below for accepted sample types, minimum volume/weight, and handling instructions. If you are working with a custom matrix or rare sample type, contact our support team for consultation.
| Sample Type | Recommended Volume / Weight | Storage Conditions | Shipping Notes |
|---|---|---|---|
| Plasma / Serum | ≥ 100–200 µL | −80°C | Ship on dry ice, avoid freeze–thaw |
| Urine | ≥ 500 µL–1 mL | −80°C | Filter or centrifuge if needed |
| Tissues (Animal/Plant) | ≥ 30–50 mg | Snap-frozen, −80°C | Pulverize if possible; ship in cryovials |
| Cultured Cells | ≥ 1–5 million cells | Pellet form, −80°C | Wash with PBS, remove media |
| Feces / Gut Microbiota | ≥ 100 mg | −80°C | Store in sterile tubes or fecal collection kits |
| CSF / Bile / Saliva | ≥ 100 µL | −80°C | Special precautions may apply (low protein samples) |
| Culture Supernatant / Media | ≥ 1 mL | −80°C | Pre-clear if possible, avoid additives |
| Soil / Environmental | ≥ 100 mg | −20°C or −80°C | Dry/freeze-dry recommended |
| Plant Extracts | ≥ 500 µL | −80°C | Record extraction solvent and method |
General Handling Notes

Metabolomics Sample Submission Guidelines
To ensure reliable and high-quality metabolomics results, proper sample handling is essential. We've prepared a comprehensive submission guide detailing:
Data Files
Quantification & Identification
Quality Control Report
Statistical & Pathway Analysis
Final Report
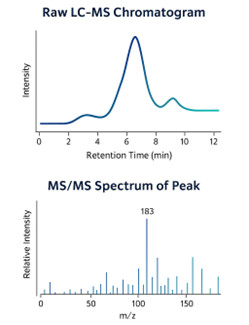
Representative LC-MS data: TIC chromatogram and MS/MS spectrum confirming metabolite ID.
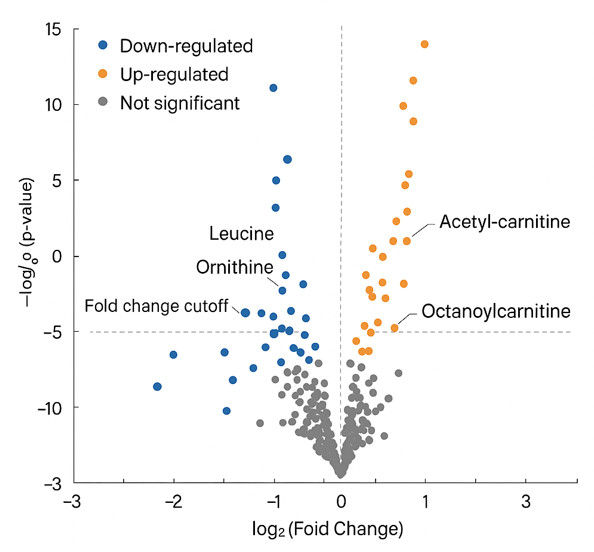
Volcano plot highlighting significantly up- and downregulated metabolites.
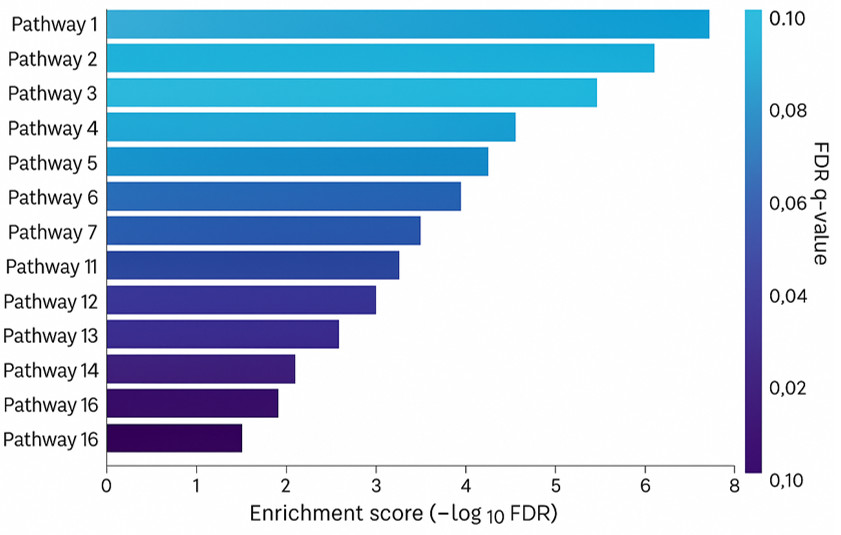
Enriched metabolic pathways ranked by significance (FDR).
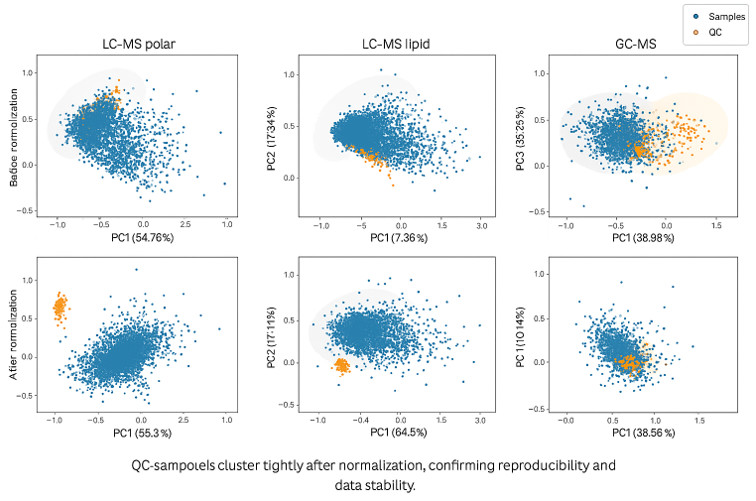
PCA plots before and after normalization. QC samples cluster tightly post-normalization.
Creative Proteomics's metabolomics service offers advanced solutions for exploring and analyzing metabolites, providing valuable insights into diverse areas of research.
Comprehensive profiling and focused study of metabolites in biofluids such blood, urine, and cerebrospinal fluid are made possible by metabolomics. Researchers can find potential biomarkers linked to particular diseases or physiological situations by examining the metabolite patterns.
Metabolomics, for instance, can assist in finding distinctive chemical signatures that distinguish between healthy people and cancer patients in cancer research. These biomarkers can help with early detection, prognosis evaluation, and therapy response tracking.


Metabolomics offers insights into drug metabolism, efficacy, and toxicity. Creative Proteomics's metabolomics service enables researchers to study the metabolic effects of drugs and identify potential drug targets. By analyzing the metabolomic profiles of cells, tissues, or biofluids after drug exposure, researchers can gain a deeper understanding of drug mechanisms of action and evaluate drug safety and efficacy.
Metabolomics can assist in identifying drug metabolites, elucidating metabolic pathways involved in drug metabolism, and predicting drug-drug interactions.
By making it easier to analyze the metabolite profiles of food samples, metabolomics enables researchers to determine the nutritional value, assess the quality of the food, and investigate how different food preparation methods affect metabolite profiles.
Understanding nutrition consumption and metabolism, finding bioactive substances in food, and assessing the impacts of additions or pollutants can all be done with the aid of metabolomics. Understanding the link between diet and health, creating individualized nutrition plans, and assuring the safety and quality of food products are all made easier with the use of this knowledge.


Metabolomics enables the analysis of metabolomic profiles in organisms exposed to environmental stressors or pollutants. By studying the metabolic responses of organisms to environmental changes, researchers can assess the impact on their metabolism and identify potential biomarkers of exposure or effect.
Metabolomics can help in understanding the biochemical pathways involved in the detoxification and metabolism of environmental contaminants. It aids in predicting ecological risks, assessing the effects of pollutants on human health, and contributing to environmental protection efforts.
Plant metabolite profiles may be analyzed more easily due to metabolomics, which reveals details about how plants use their nutrients and respond to biotic and abiotic challenges.
Understanding the biochemical pathways involved in crop development, finding metabolites linked to desired features, and optimizing agricultural techniques for better yield and quality are all possible with the help of metabolomics. This knowledge aids in the development of sustainable agricultural methods, the improvement of crop resistance to diseases, and the breeding of improved crop types.

Systems biology approaches are supported by metabolomics by providing the essential analytical tools and knowledge. Researchers can obtain a comprehensive understanding of biological processes and their regulation by integrating metabolomics data with other "omics" technologies including genomes, transcriptomics, and proteomics. Discovering new metabolic pathways, locating important regulatory nodes, and figuring out how biological phenomena are caused are all made easier by metabolomics. This holistic strategy can be used to model diseases, comprehend cellular metabolism, and find possible treatment targets.
Reference
Understanding the antecedents of cardiovascular risk in young adults is crucial for effective prevention. Traditional risk assessment methods may not capture early metabolic changes associated with cardiovascular diseases (CVD). This study aims to identify specific metabolic features in young adulthood that predict an adverse cardiovascular phenome over two decades, utilizing advanced analytical techniques.
The study includes 2330 individuals from the CARDIA cohort (mean age 32.1±3.6 years; 45% women; 45% Black). Additionally, validation is performed on 1898 participants from the FHS Offspring Cohort (mean age at metabolite measurement 54.9±9.7 years).
Statistical Methods:
Single Metabolite Regressions:
Elastic Net and PCA:
FHS Validation:
Pathway Analysis:
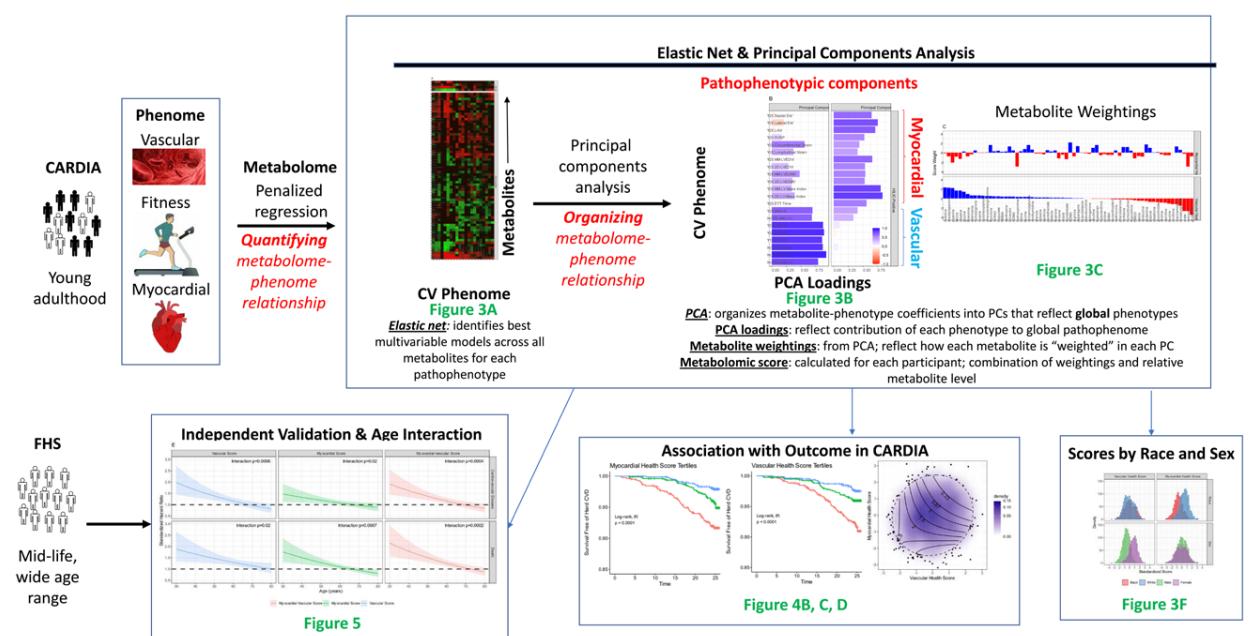 Study scheme.
Study scheme.
Clinical Characteristics:
Metabolic Dysfunction Associations:
Metabolic Scores and Cardiovascular Risk:
Validation and Age-Dependent Association:
Race Differences and Clinical Relevance:
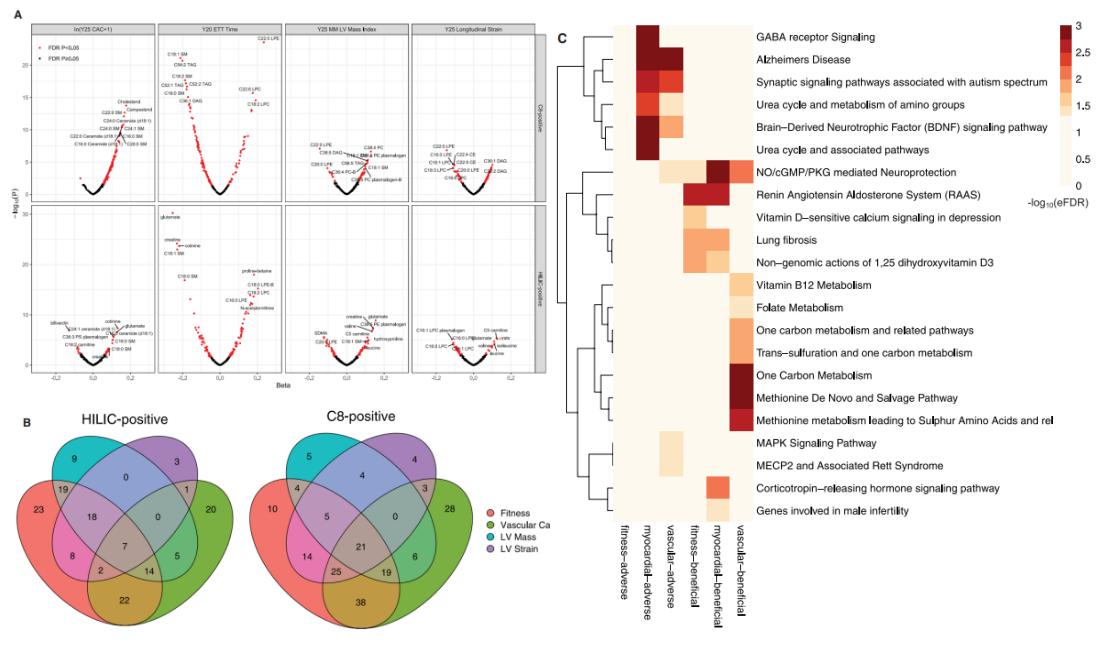 Metabolites in early adulthood are associated with the cardiovascular phenome and identify pathways central to cardiovascular disease development.
Metabolites in early adulthood are associated with the cardiovascular phenome and identify pathways central to cardiovascular disease development.
Reference
What are external standards and internal standards in metabolomics? If an internal standard is added during detection, does that constitute internal standard method detection?
External standards refer to standard substances used in metabolite detection that are consistent with the analyte. Since the concentration and amount of the standard substance are known, by using standard substances of different concentrations to construct a standard curve and obtain peak areas, the concentration of the analyte can be calculated based on the peak areas of the standard curve, the concentration of the standard curve, and the peak area of the analyte, thereby achieving absolute quantification and qualitative analysis of the analyte.
Internal standards refer to isotopes of the analyte used in metabolite detection. Isotopes are detected together with the analyte. Since the concentration and amount of isotopes are known, the content of the analyte can be inferred based on the peak areas of the isotopes and the analyte, thereby achieving relative quantification and qualitative analysis of the analyte. Adding an internal standard does not necessarily constitute internal standard method detection. External standards are used for absolute quantification and qualitative analysis of the analyte, while internal standards are used to correct quantitative differences caused by variations in sample amounts and eliminate the influence of matrices on quantification.
What are the requirements for biological replicates in metabolomics?
Compared to other omics studies, metabolomics, being downstream, requires a larger number of samples to avoid analytical errors due to individual differences in metabolic profiles. It is generally recommended to have at least 8 parallel samples for plant, microbial, and cellular samples, around 10 parallel samples for model animals, and at least 30 parallel samples for clinical samples. If the number of biological replicates is too small, the subsequent model construction will be poor, and there will be very few differentially expressed metabolites that meet the conditions.
If the sample quality is below the collection standard, can metabolomics analysis still be performed?
Because the quality of samples in metabolomics analysis is often positively correlated with the number of metabolites detected, the requirement of 100mg in the collection standard is based on project experience, balancing the customized sample requirements and the quantity of detected substances. It does not mean that samples below 100mg cannot be tested; it just indicates that fewer substances might be detected compared to standard samples of the same type.
For clinical samples with a long collection period, can they be batched for testing and combined analysis?
It is not recommended to batch test and combine analysis as different instrument states at different times can affect the results and even change the number of detected differential molecules. It is advisable to test all samples together after collection. If the sample size is large and it's not possible to test all samples in one batch, we have strategies for batch testing large sample queues to minimize batch effects.
Can untargeted metabolomics analyze different tissue types together (e.g., one being animal tissue and the other intestinal contents)?
They can be analyzed together on the same machine and analyzed together using shared quality controls (QCs). If you don't want to mix a QC, and want to use two untargeted results for combined analysis, theoretically, we do not recommend it because different tissue types contain different types of substances, and some substances specific to certain tissue types might be filtered out during data filtering.
What concentration of extracted metabolites is required for machine analysis?
Extracted samples can undergo untargeted metabolomics machine analysis; the approximate concentration of the extracted material needs to be noted. Additionally, certain extraction agents (e.g., DMSO) may crystallize at low temperatures, so special notes are necessary. If the extraction concentration is too high, dilution might be needed. The laboratory typically uses methanol for dilution, and the maximum concentration for machine analysis is 20ppm.
Does the laboratory perform desalination for high-salt samples?
A: The laboratory directly performs metabolite extraction without desalination steps. High salt content can interfere with metabolite detection, and customers are informed of this risk beforehand. The laboratory will rely on actual test results, and any anomalies during the experiment will be promptly reported.
Why aren't untargeted results validated by targeted methods?
Non-targeted and targeted metabolite detection are two completely different principles. Non-targeted metabolomics is broad-spectrum, aiming to collect as many metabolite ion peaks as possible through different chromatographic columns and ionization methods, identifying differential metabolites through database comparisons to find biomarkers or compare differences between different groups. Targeted metabolomics, on the other hand, quantitatively analyzes and compares target metabolites using Multiple Reaction Monitoring (MRM) methods, matching parent ion/daughter ion pairs. Thus, the two methods have different purposes, identification methods, and result presentations. Additionally, the extraction methods differ: untargeted extraction methods obtain most metabolites from samples without specificity, while targeted methods are specific to certain types of substances.
Can untargeted metabolomics data determine the presence or absence of substances?
After each sample is run separately, all mass spectrometry files of the samples are processed together in software like XCMS for deconvolution, peak alignment, retention time correction, and null value filling. After merging and searching the library for qualitative analysis, differences between different samples are eliminated. Subsequently, based on the principle of >50%, metabolites are selected, with substances appearing less than 50% being removed, and the remaining substances are filled with null values. Therefore, there is no concept of presence or absence in metabolomics results.
What is the difference between metabolic ions and metabolites?
Metabolic ions detected in mass spectrometers are annotated during data analysis, resulting in metabolites. Results annotated using mass-to-charge ratio are primary metabolites, while those annotated using peak spectrum are secondary metabolites. Therefore, the results of secondary metabolites are more accurate than those of primary metabolites and are preferred for analysis.
What do "pos" and "neg" mean?
A: "Pos" and "neg" represent two scanning modes. After ionization, some metabolites tend to carry a positive charge, while others tend to carry a negative charge. To detect more metabolite ions, we perform scanning in both positive ion mode and negative ion mode. For subsequent data filtering, it is recommended to analyze the results of both modes to obtain a more complete result.
MS-CETSA functional proteomics uncovers new DNA-repair programs leading to Gemcitabine resistance
Nordlund, P., Liang, Y. Y., Khalid, K., Van Le, H., Teo, H. M., Raitelaitis, M., ... & Prabhu, N.
Journal: Research Square
Year: 2024
High Levels of Oxidative Stress Early after HSCT Are Associated with Later Adverse Outcomes
Cook, E., Langenberg, L., Luebbering, N., Ibrahimova, A., Sabulski, A., Lake, K. E., ... & Davies, S. M.
Journal:Transplantation and Cellular Therapy
Year: 2024
Multiomics of a rice population identifies genes and genomic regions that bestow low glycemic index and high protein content
Badoni, S., Pasion-Uy, E. A., Kor, S., Kim, S. R., Tiozon Jr, R. N., Misra, G., ... & Sreenivasulu, N.
Journal: Proceedings of the National Academy of Sciences
Year: 2024
The Brain Metabolome Is Modified by Obesity in a Sex-Dependent Manner
Norman, J. E., Milenkovic, D., Nuthikattu, S., & Villablanca, A. C.
Journal: International Journal of Molecular Sciences
Year: 2024
UDP-Glucose/P2Y14 Receptor Signaling Exacerbates Neuronal Apoptosis After Subarachnoid Hemorrhage in Rats
Kanamaru, H., Zhu, S., Dong, S., Takemoto, Y., Huang, L., Sherchan, P., ... & Zhang, J. H.
Journal: Stroke
Year: 2024
Pan-lysyl oxidase inhibition disrupts fibroinflammatory tumor stroma, rendering cholangiocarcinoma susceptible to chemotherapy
Burchard, P. R., Ruffolo, L. I., Ullman, N. A., Dale, B. S., Dave, Y. A., Hilty, B. K., ... & Hernandez-Alejandro, R.
Journal: Hepatology Communications
Year: 2024
Comparative metabolite profiling of salt sensitive Oryza sativa and the halophytic wild rice Oryza coarctata under salt stress
Tamanna, N., Mojumder, A., Azim, T., Iqbal, M. I., Alam, M. N. U., Rahman, A., & Seraj, Z. I.
Journal: Plant‐Environment Interactions
Year: 2024
Teriflunomide/leflunomide synergize with chemotherapeutics by decreasing mitochondrial fragmentation via DRP1 in SCLC
Mirzapoiazova, T., Tseng, L., Mambetsariev, B., Li, H., Lou, C. H., Pozhitkov, A., ... & Salgia, R.
Journal: iScience
Year: 2024
Physiological, transcriptomic and metabolomic insights of three extremophyte woody species living in the multi-stress environment of the Atacama Desert
Gajardo, H. A., Morales, M., Larama, G., Luengo-Escobar, A., López, D., Machado, M., ... & Bravo, L. A.
Journal: Planta
Year: 2024
A personalized probabilistic approach to ovarian cancer diagnostics
Ban, D., Housley, S. N., Matyunina, L. V., McDonald, L. D., Bae-Jump, V. L., Benigno, B. B., ... & McDonald, J. F.
Journal: Gynecologic Oncology
Year: 2024
Glucocorticoid-induced osteoporosis is prevented by dietary prune in female mice
Chargo, N. J., Neugebauer, K., Guzior, D. V., Quinn, R. A., Parameswaran, N., & McCabe, L. R.
Journal: Frontiers in Cell and Developmental Biology
Year: 2024
Proteolytic activation of fatty acid synthase signals pan-stress resolution
Wei, H., Weaver, Y. M., Yang, C., Zhang, Y., Hu, G., Karner, C. M., ... & Weaver, B. P.
Journal: Nature Metabolism
Year: 2024
Quantifying forms and functions of intestinal bile acid pools in mice
Sudo, K., Delmas-Eliason, A., Soucy, S., Barrack, K. E., Liu, J., Balasubramanian, A., … & Sundrud, M. S.
Journal: bioRxiv
Year: 2024
Elevated SLC7A2 expression is associated with an abnormal neuroinflammatory response and nitrosative stress in Huntington's disease
Gaudet, I. D., Xu, H., Gordon, E., Cannestro, G. A., Lu, M. L., & Wei, J.
Journal: Journal of Neuroinflammation
Year: 2024
Thermotolerance capabilities, blood metabolomics, and mammary gland hemodynamics and transcriptomic profiles of slick-haired Holstein cattle during mid lactation in Puerto Rico
Contreras-Correa, Z. E., Sánchez-Rodríguez, H. L., Arick II, M. A., Muñiz-Colón, G., & Lemley, C. O.
Journal: Journal of Dairy Science
Year: 2024
DNA stimulates SIRT6 to mono-ADP-ribosylate proteins within histidine repeats
Pederson, N. J., & Diehl, K. L.
Journal: bioRxiv
Year: 2024
Glycine supplementation can partially restore oxidative stress-associated glutathione deficiency in ageing cats
Ruparell, A., Alexander, J. E., Eyre, R., Carvell-Miller, L., Leung, Y. B., Evans, S. J., ... & Watson, P.
Journal: British Journal of Nutrition
Year: 2024
Untargeted metabolomics reveal sex-specific and non-specific redox-modulating metabolites in kidneys following binge drinking
Rafferty, D., de Carvalho, L. M., Sutter, M., Heneghan, K., Nelson, V., Leitner, M., ... & Puthanveetil, P.
Journal: Redox Experimental Medicine
Year: 2023
Sex modifies the impact of type 2 diabetes mellitus on the murine whole brain metabolome
Norman, J. E., Nuthikattu, S., Milenkovic, D., & Villablanca, A. C.
Journal: Metabolites
Year: 2023
A human iPSC-derived hepatocyte screen identifies compounds that inhibit production of Apolipoprotein B
Liu, J. T., Doueiry, C., Jiang, Y. L., Blaszkiewicz, J., Lamprecht, M. P., Heslop, J. A., ... & Duncan, S. A.
Journal: Communications Biology
Year: 2023
Methyl donor supplementation reduces phospho‐Tau, Fyn and demethylated protein phosphatase 2A levels and mitigates learning and motor deficits in a mouse model of tauopathy
van Hummel, A., Taleski, G., Sontag, J. M., Feiten, A. F., Ke, Y. D., Ittner, L. M., & Sontag, E.
Journal: Neuropathology and Applied Neurobiology
Year: 2023
Sex hormones, sex chromosomes, and microbiota: identification of Akkermansia muciniphila as an estrogen-responsive bacterium
Sakamuri, A., Bardhan, P., Tummala, R., Mauvais-Jarvis, F., Yang, T., Joe, B., & Ogola, B. O.
Journal: Microbiota and Host
Year: 2023
Living in extreme environments: a photosynthetic and desiccation stress tolerance trade-off story, but not for everyone
Gajardo, H. A., Morales, M., López, D., Luengo-Escobar, M., Machado, A., Nunes-Nesi, A., ... & Bravo, L.
Journal: Authorea Preprints
Year: 2023
Resting natural killer cell homeostasis relies on tryptophan/NAD+ metabolism and HIF‐1α
Pelletier, A., Nelius, E., Fan, Z., Khatchatourova, E., Alvarado‐Diaz, A., He, J., ... & Stockmann, C.
Journal: EMBO Reports
Year: 2023
Function and regulation of a steroidogenic CYP450 enzyme in the mitochondrion of Toxoplasma gondii
Asady, B., Sampels, V., Romano, J. D., Levitskaya, J., Lige, B., Khare, P., ... & Coppens, I.
Journal: PLoS Pathogens
Year: 2023

Explore the future of metabolomics with Creative Proteomics. Discover cutting-edge solutions for metabolic analysis and research.
Copyright © 2026 Creative Proteomics. All rights reserved.