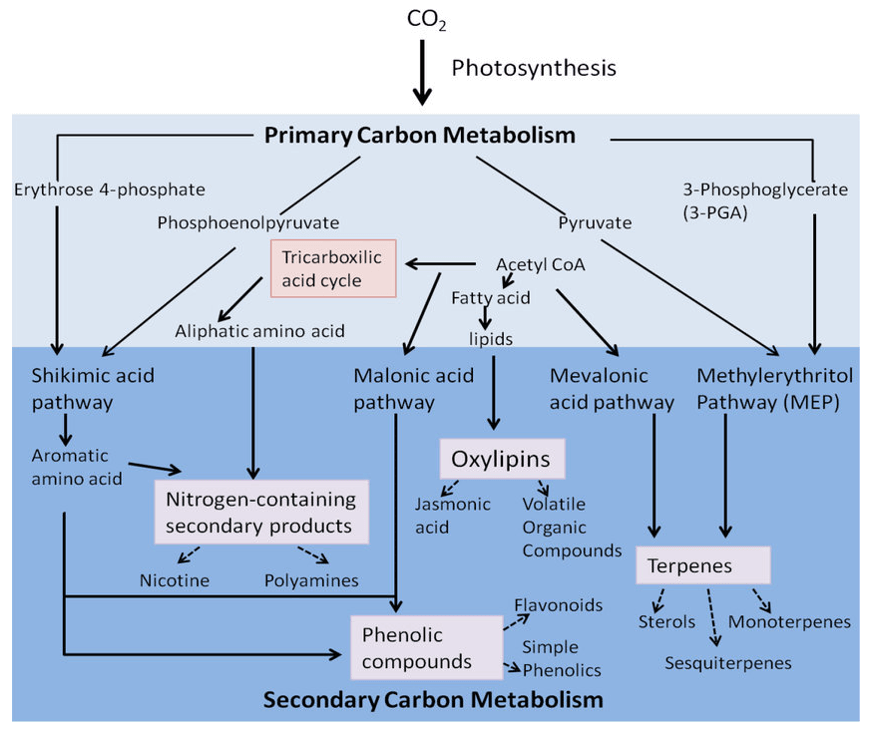
Concanavalin Analysis Service
Submit Your InquiryConcanavalin A (ConA) is a lectin (carbohydrate binding protein) originally extracted from Canavalia ensiformis. Concanavalin binds specifically to certain structures in various sugars, glycoproteins and glycolipids, mainly internal and non-reducing terminal α-D-mannosyl and α-D-glucosyl groups. ConA is the first commercially available lectin and is widely used in various studies to characterize glycoproteins and other sugar-containing entities on the surface of various cells. ConA has a broad-spectrum antiviral effect and also has a therapeutic effect on bacterial diseases. Concanavalin can also activate cells in a sick state, and can obviously treat some immunodeficiency diseases, as well as other severe diseases caused by bacterial or viral mixed infections. Based on LC-MS technology, Creative Proteomics offers qualitative and quantitative analysis of concanavalin A to meet your needs.
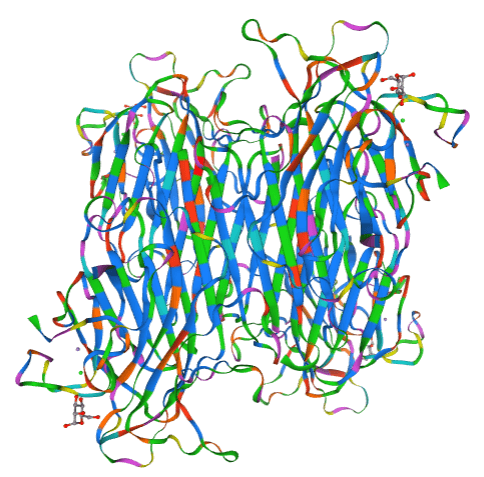 Schematic diagram of ConA structure
Schematic diagram of ConA structure
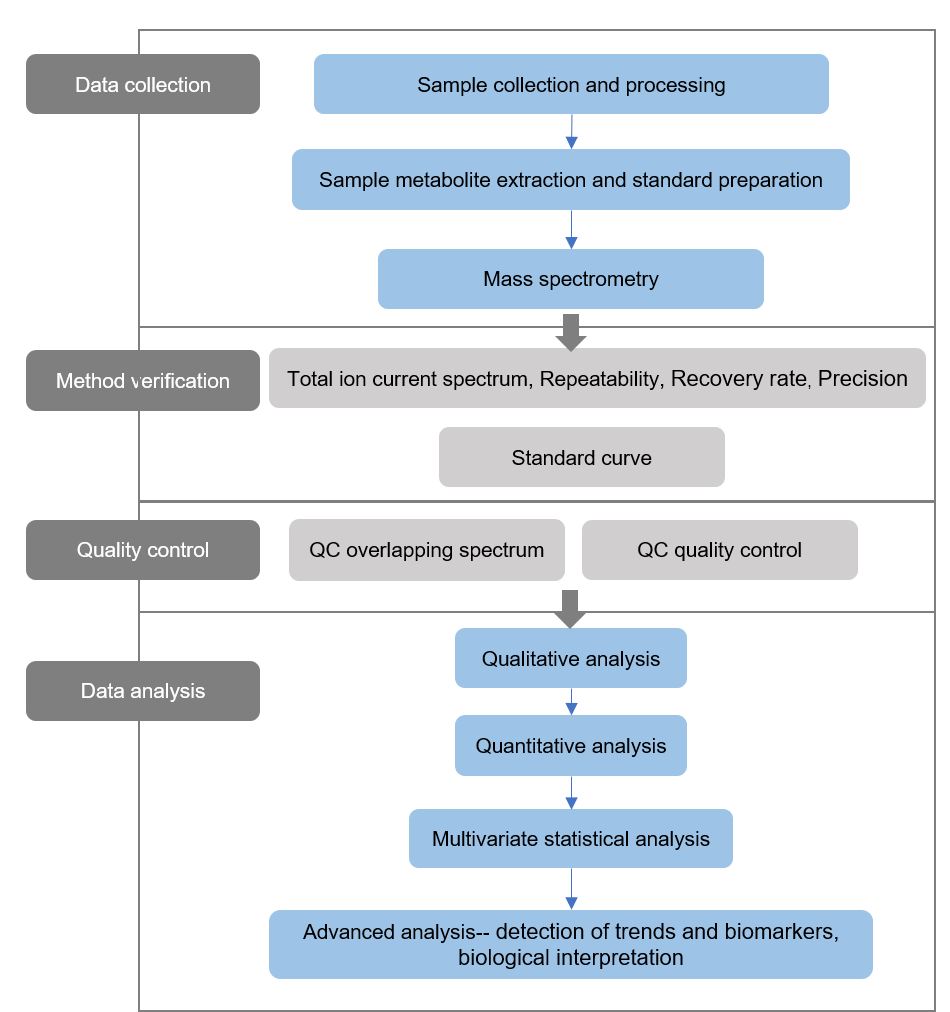
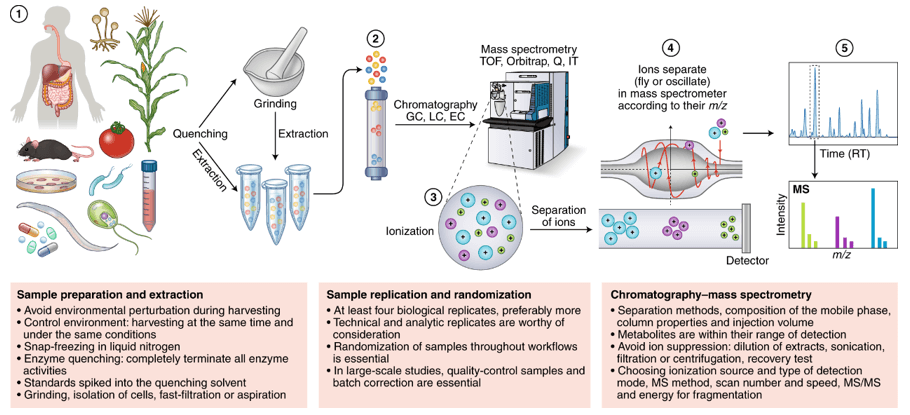
Technical Route of Concanavalin (ConA) Analysis
Sample Preparation
Before subjecting Con A to LC-MS analysis, it is essential to prepare the sample appropriately. Typically, this involves the extraction and purification of Con A from the source material. Various techniques, such as affinity chromatography using α-D-mannose or α-D-glucose affinity columns, can be employed to isolate Con A with high purity.
LC Separation
Once the Con A sample is prepared, it can be injected into an LC system for separation. Reverse-phase chromatography (RPC) is commonly used for Con A analysis. In RPC, a nonpolar stationary phase and a polar mobile phase are employed to separate molecules based on their hydrophobicity. The choice of column and mobile phase composition may vary depending on the specific experimental requirements.
MS Detection
After separation by LC, the Con A molecules are introduced into the mass spectrometer for detection. The mass spectrometer ionizes the analyte molecules and separates them based on their mass-to-charge ratio (m/z). The most commonly used ionization techniques for Con A analysis include electrospray ionization (ESI) and matrix-assisted laser desorption/ionization (MALDI).
ESI is a soft ionization technique that generates multiply charged ions, allowing accurate mass determination and fragmentation studies. MALDI, on the other hand, is a laser-based ionization method that is particularly suitable for analyzing large biomolecules. Both techniques offer unique advantages and can be chosen based on the specific requirements of the Con A analysis.
Data Analysis
The data obtained from the LC-MS analysis of Con A is typically processed and analyzed using specialized software tools. These tools enable the identification of Con A based on its mass and charge properties, as well as the characterization of any post-translational modifications or glycosylation patterns present.
Applications of Con A Analysis
The analysis of Con A using LC-MS platform can be applied to:
Glycoprotein Analysis: Con A is widely used for the enrichment and analysis of glycoproteins. By exploiting its carbohydrate-binding properties, Con A can selectively isolate glycoproteins from complex biological samples, allowing for their subsequent characterization by LC-MS.
Glycomics Profiling: LC-MS analysis of Con A can be utilized to study the glycomics profiles of biological samples. By identifying and quantifying the glycan structures bound by Con A, researchers can gain insights into the glycosylation patterns of proteins and their potential roles in various biological processes.
Biopharmaceutical Analysis: LC-MS analysis of Con A can be applied in the quality control and characterization of biopharmaceutical products, particularly those derived from mammalian cell cultures. It allows for the detection and quantification of glycosylation variants, which can impact the efficacy and safety of therapeutic proteins.
Biomarker Discovery: Con A analysis by LC-MS can contribute to biomarker discovery efforts. By comparing the glycosylation profiles of different biological samples, researchers can identify potential glycoprotein biomarkers associated with specific diseases or physiological conditions.
What You May Want to Know About Con A Analytics Platform
Detection Sensitivity and Limit
The detection sensitivity of Con A analysis depends on the detection techniques and the sensitivity of the instrument used. Common LC-MS systems typically have good sensitivity and can detect Con A at sub-picomolar levels. However, the specific detection limit can be influenced by sample preparation and quality. We will optimize sample handling to reduce interference and improve detection sensitivity and detection limits.
Selection of Suitable Ionization Techniques
Common ionization techniques used in Con A analysis include electrospray ionization (ESI) and matrix-assisted laser desorption/ionization (MALDI). ESI is suitable for solution samples, providing high ion yields and good quantification capabilities. On the other hand, MALDI is suitable for solid samples and analysis of large molecules, offering high ionization efficiency and the ability to acquire mass spectra images.
Identification and Characterization Methods
The identification of Con A is typically based on its mass spectra and characteristic mass-to-charge ratio (m/z) values. Mass spectra can be obtained using LC-MS instruments and compared with known Con A spectra. Various software tools and databases can assist in the identification and characterization of Con A. Additionally, other techniques like tandem mass spectrometry (MS/MS) can provide structural information about Con A and its binding partners.
Data Analysis and Quantification
Con A analysis data can be analyzed using dedicated software packages designed for LC-MS data processing. These tools allow for peak integration, deconvolution, and quantification of Con A and its interacting molecules. Quantification can be achieved by comparing the peak areas or intensities with internal or external standards. Additionally, statistical analysis methods can be applied to ensure the accuracy and reliability of the quantification results.