Globo- and Isoglobo- Series of Glycosphingolipid Analysis Service
Submit Your InquiryOverview
Glycosphingolipids (GSLs) were discovered by the German chemist Johannes Thudichum in the late 19th century when he analysed the components of the human brain in his laboratory in London. Thudichum carefully identified the structure of novel lipids encountered, consisting of distinct lipid moieties with linked glycan or phosphocholine groups. In vertebrates, glycosphingolipids have great structural heterogeneity, and more than 300 different polysaccharides have been identified to date.
In cells, molecular synthesis of GSL starts in the endoplasmic reticulum (ER), where serine palmitoyl transferase (SPT) generates keto-sphingosine from serine and palmitoyl -CoA. They are classified into several subfamilies, including the ganglio-, globo-, and neolacto-series of GSLs. Various glycan structures on glycosphingolipids have been found to be important for molecular recognition. Viral and pathogenic bacterial adhesin utilize glycosphingolipids on that surface of host cell to bind and invade epithelial cells, and the binding is microbially specific for glycan structures. In addition to being a key component of cell recognition, structurally diverse glycosphingolipids with different glycan structures are also involved in cell signaling.
Creative Proteomics, as the leader of bio-tech industry, devote to forge high quality targeted metabolomics service with advanced apparatus and professional research team. We provide high throughput, high precision and short time-consuming Globo- and Isoglobo- Series of Glycosphingolipid Analysis Service, which could be meet all the need of our customers, to assist your scientific research.
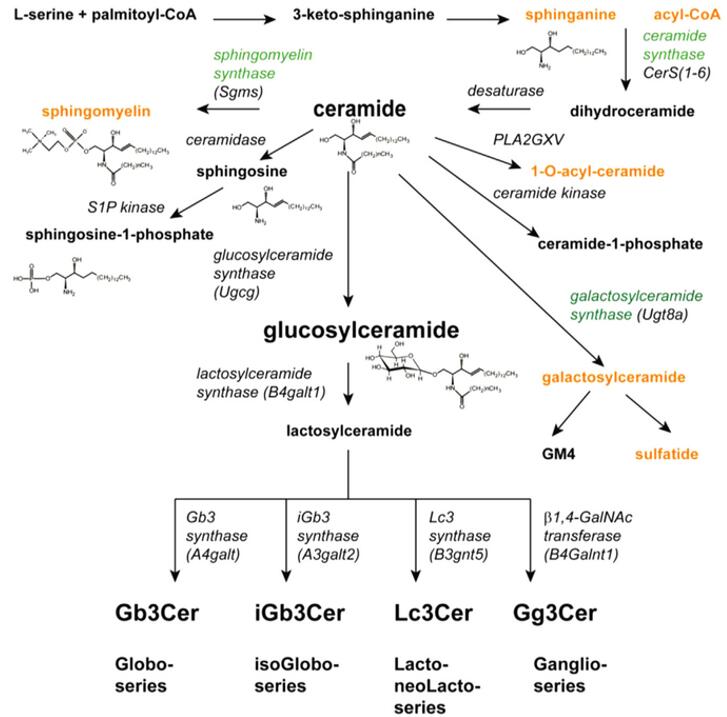 Fig 1. Pathways for sphingolipid synthesis.
Fig 1. Pathways for sphingolipid synthesis.
Advantages
- Short time-consuming
- High sensitivity and few detection restrictions
- High precision and good repeatability
- High throughput
- Customized service
Service workflow
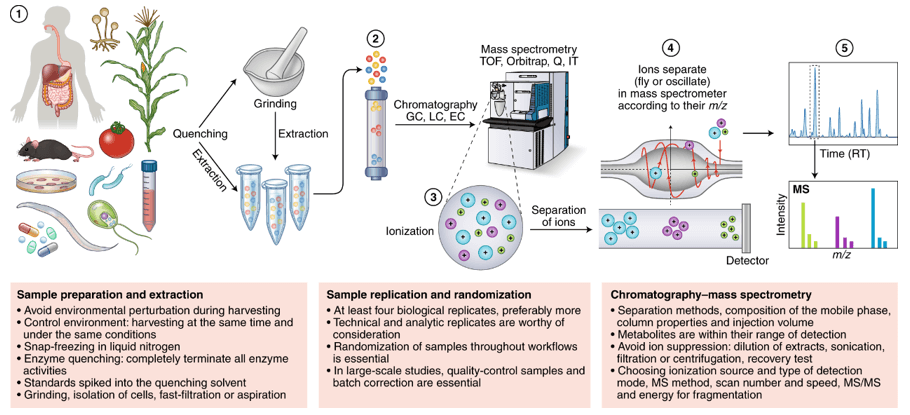
- Extract and purifying glycosphingolipid from cell membrane or serum
- Release that glycosphingolipid glycan with the glucosidase
- The total methylation of glycan
- UPLC-HILIC-FLD analysis based on 2AB labeling
- Analysis of fully methylated glycans by time-of-flight mass spectrometry
 Fig 2. Workflow of common Glycosphingolipids analysis in various type of animal samples.
Fig 2. Workflow of common Glycosphingolipids analysis in various type of animal samples.
Sample requirement
- Serum/plasma: 500 μl/sample
- Protein: 100 µg
- Anticoagulated blood (EDTA): 1 mL
- Urine: 1 ml/sample
- Animal tissues: 200 mg/sample
- Cells: ≥ 1 × 107/sample
- Feces: 500 mg/sample
Repeated freezing and thawing of samples must be avoided. The serum sample should be precipitated in the collection tube for 30 minutes at room temperature, then transported to the centrifuge tube and centrifuged at 8000 rpm for 5 minutes. After centrifugation, the supernatant was equally divided into a freezing tube of 500 uL / sample.
Anticoagulants and preservatives must be added immediately after collection and then frozen at -80 °C.
Urine samples should be equally divided into centrifuge tubes with 1 mL per tube, each tube is added with 1/100 (w/v) sodium azide and stored at -80 °C.
Samples should be frozen in liquid nitrogen immediately and then transported to -80 °C for storage after collected.
Cytoactive should be terminated immediately to maintaining cell integrity.
In general, to assure enough sample to fulfill the whole project, the volume of the single sample need to be offered as much as possible. The remaining samples will be stored for one year free of charge and returned at any time if necessary. All samples need to be stored and transported at -80°C and try to avoid using surfactants (SDS, Triton-X) and inorganic salts.
Clinical samples are repeated in no less than 30 cases in a single group.
Animal samples are repeated in no less than 9 cases in a single group.
Report delivery
- Experimental procedure
- Parameters of HPLC and MS
- Raw data, chromatograms and mass spectra
- Metabolites quantification data
- Custom analysis report
Service cycle
- Sample testing: 5-10 working days
- Data analysis: 5-10 working days
Creative Proteomics metabolism analysis platform is committed to the all-around, reliable and accurate analysis service for a variety of target substances, which is suitable for life-science research, drug exploration, biological determination and other fields. We sincerely hope to cooperate with you to assistant your scientific research.
References
- Z. Wang, K. Arnold, Y. Xu, et al. Quantitative analysis of heparan sulfate using isotopically labeled calibrants. Commun Biol, 2020, 3(1):425.
- H. Yu, Y. Li, J. Zeng, et al. Sequential One-Pot Multienzyme Chemoenzymatic Synthesis of Glycosphingolipid Glycans. J Org Chem, 2016, 81(22):10809-10824.