Salvianic Acid Analysis Service
Submit Your InquiryOverview
Salvianic acid is a kind of organic compound, which is a phenolic aromatic acid first isolated from panax japonica ginseng. Salvianic acid is a brownish-yellow powder or yellow powder, and its sodium salt is a white needle-like crystal, which is one of the main medicinal ingredients in water-soluble components of salvia miltiorrhiza. Salvianic acid has no detectable rotation in ethanol. It can react with FeCl3, and the solution after the reaction is yellow-green. Salvianic acid has characteristic infrared spectrum and nuclear magnetic resonance spectrum, which is helpful for its identification and analysis. Clinically, salvianic acid has certain effects on the heart, coronary circulation, microcirculation, blood coagulation and platelet aggregation. It has the effects of promoting blood circulation to remove blood stasis, and relieving pain, which can be used for chest tightness and angina pectoris. Because salvianic acid has a wide range of uses, it is very important to establish the method for the analysis of salvianic acid in plant metabolites. Creative Proteomics provides the simple, rapid, accurate, affordable and highly automated analytical method and equipment to analyze salvianic acid. High performance liquid chromatography (HPLC) can solve some of the problems associated with salvianic acid analysis. This analysis method is very simple, fast, accurate and reproducible. Improved extraction and purification steps can also increase the sensitivity of the analytical method.
Applications of Salvianic Acid Analysis
- Qualitative analysis of the main types of salvianic acid
- Quantitative analysis of salvianic acid content
- Ensure safety and quality
- Provide basis for improvement
Advantages of Our Salvianic Acid Analysis Service
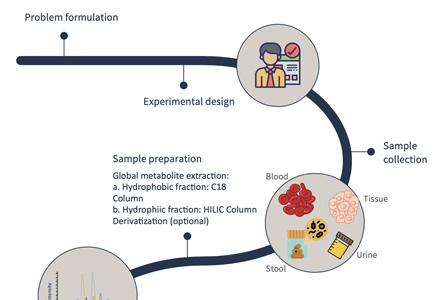
Service Workflow
Our newly developed sample preprocessing method provides safety, environmental protection, rapidness, effectiveness, and the average recovery rate is up to 99%. Sample preprocessing at Creative Proteomics complies with the in-house standard operation procedures (SOP), ensuring maximized homogeneity between samples.
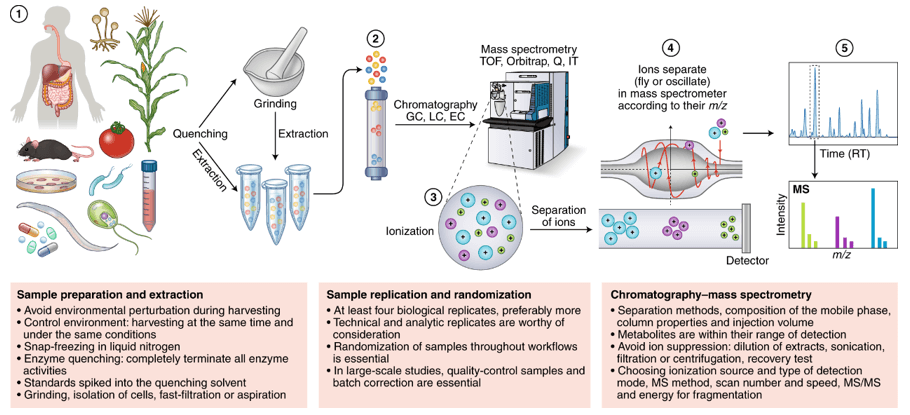
 Figure 1. Salvianic acid analysis service workflow.
Figure 1. Salvianic acid analysis service workflow.
Methods: RP - HPLC method
Mobile phase: methanol-glacial acetic acid
Detector: UV detector
Linear range: r>0.999
Time: dozens of minutes, slightly different according to different ingredients
Analysis content:
- Standard curve creation
- Raw data preprocessing
- Absolute quantitation of salvianic acid
- Differential metabolite screening
- Optimal analyses
Sample Requirements
1. Fresh tissues >1 g. If possible, samples should be frozen in liquid nitrogen right after collection, and then transferred to -80°C for storage.
2. Product samples.
At least 3 biological replicates.
Delivery
- Experimental procedure
- Parameters of HPLC
- Purity analysis report
- HPLC raw data files and HPLC data quality checks
- Metabolites quantification data
- Custom analysis report
HPLC based profiling of salvianic acid enables quantitative analyses in a faster, more convenient, and more sensitive manner. With decades of experience in HPLC services, Creative Proteomics has a proven track record supporting diverse salvianic acid detection and quantitation. We can meet your specific project requirements, from sample to bioinformatics.