Ascorbate and Aldarate Metabolism Service
Submit Your InquiryWhy Analyze Ascorbate and Aldarate Metabolism?
Ascorbate and aldarate metabolism is one of important carbohydrate metabolic pathways and can protect cells against oxidative damage resulting from aerobic metabolism and a range of pollutants. Ascorbate and aldarate metabolism likely affect the oxidative stress response which is closely involved in transformation of L-urobilinogen from L-Urobilin, both of which is related to the oxidative stress. The oxidative stress stimulates the generation of reactive oxygen species, which is closely associated to a lot of pathologies of diseases and disorders such as cardiovascular disease, cancer, aging, and various neurodegenerative diseases. It would not be surprising that aberrant ascorbate and aldarate metabolism links to various diseases and disorders by interfering with oxidative stress. As a high-throughput tool, ascorbate and aldarate metabolism analysis provides a powerful way to diagnosis and gain new insights in disease mechanism or treatment. With integrated array of separation, characterization, identification and quantification experience, Creative Proteomics provides reliable, rapid and cost-effective ascorbate and aldarate metabolism analysis service for your various scientific purposes.
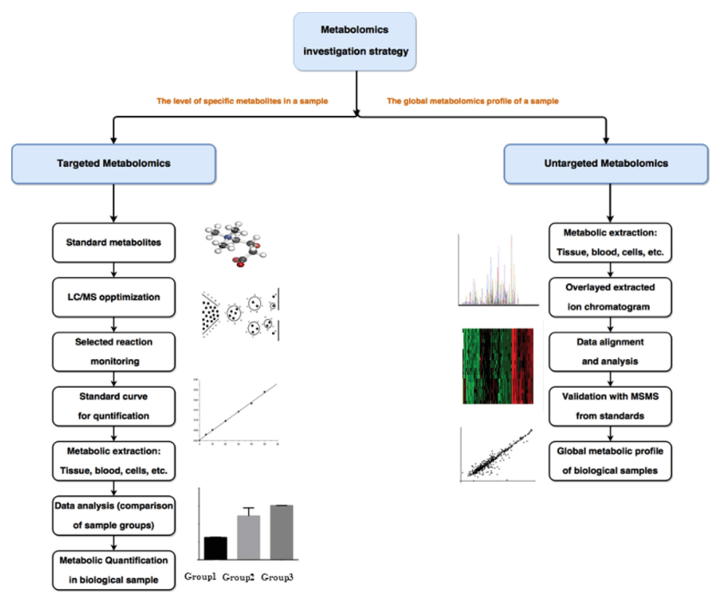
Workflow of Ascorbate and Aldarate Metabolism
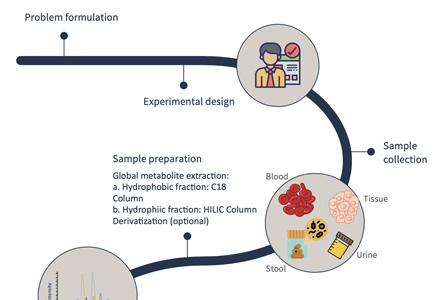
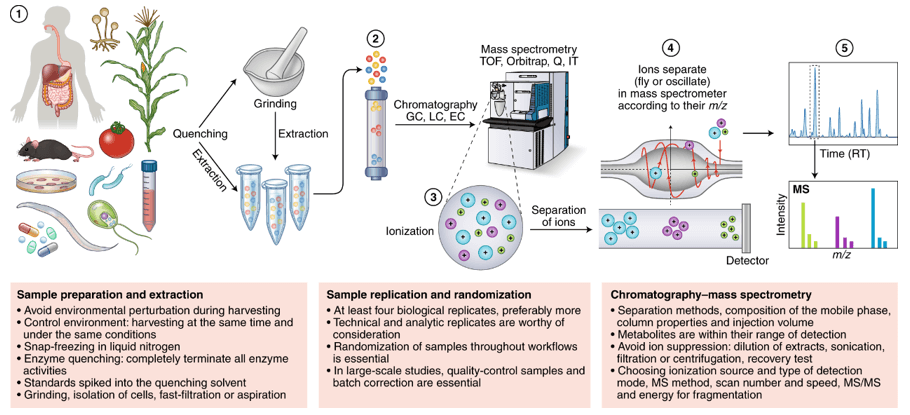
Sample Collection and Preparation: Obtain biological samples, such as tissues, cells, or biofluids, and carefully prepare them for analysis. Sample preparation techniques involve extraction, fractionation, and derivatization to improve metabolite stability and enhance detection.
Analytical Techniques: Employ advanced analytical techniques to detect, separate, and quantify the metabolites. Liquid chromatography-mass spectrometry (LC-MS) and gas chromatography-mass spectrometry (GC-MS) are commonly utilized for their high sensitivity, specificity, and broad coverage of metabolites.
Data Acquisition: Acquire raw data from the chosen analytical platforms. LC-MS and GC-MS generate complex data sets consisting of mass spectra and retention times, which represent the metabolite profiles in the samples.
Data Preprocessing: Perform data preprocessing steps to enhance data quality and reduce noise. This involves data alignment, peak picking, deconvolution, and normalization to facilitate accurate comparisons between samples.
Metabolite Identification: Apply various strategies for metabolite identification, including spectral matching against reference libraries, tandem mass spectrometry (MS/MS) fragmentation, and isotopic labeling experiments. These techniques aid in annotating metabolites and confirming their identities.
Statistical Analysis: Perform statistical analysis to identify significant differences between sample groups, such as control vs. treatment or healthy vs. diseased. Multivariate statistical methods, such as principal component analysis (PCA) and partial least squares-discriminant analysis (PLS-DA), enable the detection of metabolic alterations associated with ascorbate and aldarate metabolism.
Pathway Analysis: Utilize pathway analysis tools to map identified metabolites onto metabolic pathways. This analysis provides a holistic view of the interconnected metabolic network associated with ascorbate and aldarate metabolism.
Biomarker Discovery: Identify potential biomarkers or metabolite signatures associated with specific conditions or disease states. Differential metabolite analysis combined with machine learning algorithms allows for the discovery of discriminatory metabolites.
 Figure 1. The overall workflow of ascorbate and aldarate metabolites service.
Figure 1. The overall workflow of ascorbate and aldarate metabolites service.
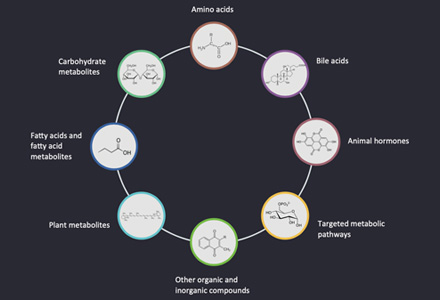
List of Detectable Ascorbate and Aldarate Metabolism at Creative Proteomics
| Dehydroascorbic acid | 2,3-diketo-L-gulonate | L-lyxonate |
| L-erythrulose | Oxalate | Threonate |
| Oxidized glutathione |
Sample Requirements for Ascorbate and Aldarate Metabolism Analysis
Biological Samples: Biological samples commonly used for ascorbate and aldarate metabolism analysis include:
a. Tissues: Tissue samples, such as liver, kidney, brain, or muscle, can provide insights into the metabolic processes occurring in specific organs. Proper tissue collection, preservation, and storage are essential to maintain metabolite stability. Snap-freezing in liquid nitrogen or cryopreservation at ultra-low temperatures (-80°C) are common preservation methods.
b. Cells: Cell cultures, both primary and cell lines, allow for controlled experiments and mechanistic studies. Cells can be harvested using standard techniques like trypsinization or scraping, and subsequent metabolite extraction can be performed using appropriate methods.
c. Biofluids: Biofluids such as blood, plasma, serum, urine, or cerebrospinal fluid (CSF) can provide a systemic perspective on ascorbate and aldarate metabolism. Care should be taken during collection to prevent contamination or degradation of metabolites. Anticoagulants or preservatives, such as heparin or EDTA, may be required for blood sample collection.
Sample Collection and Storage: Proper sample collection, handling, and storage are crucial to maintain the integrity of metabolites. Samples should be collected under appropriate conditions to minimize degradation or alterations. For long-term storage, samples should be stored at ultra-low temperatures (-80°C or below) to preserve metabolite stability. It is advisable to avoid multiple freeze-thaw cycles, as this can lead to metabolite degradation.
Preventing Contamination: To avoid contamination, it is essential to use sterile collection and storage containers and ensure proper aseptic techniques during sample collection. Contamination can introduce external metabolites or alter the metabolic profile, leading to inaccurate results.
Report Delivery
- Experimental procedures
- Instrument parameters of liquid chromatography and mass spectrometry
- MS raw data files and the summary of mass spectrometry data quality
- The identification/quantification of lipidic molecules data
- Bioinformatics analysis report (PCA, KEGG, etc.)
Equipped with advanced mass spectrometry for the determination of ascorbate and aldarate metabolism, powerful bioinformatic analysis software and a panel of experienced technicians and scientists, we provide customer-tailored ascorbate and aldarate metabolism analysis service with rapid experimental procedures and easy to read report, to advance your scientific research.
References
- Sun Lili, Jia Hongmei, Li Jiaojiao, et al. Cecal Gut Microbiota and Metabolites Might Contribute to the Severity of Acute Myocardial Ischemia by Impacting the Intestinal Permeability, Oxidative Stress, and Energy Metabolism. Front Microbiol. 2019, 10:1745.
- Brieger K, Schiavone S, Miller FJ, Krause K-H. Reactive oxygen species: from health to disease. Swiss Med Wkly. 2012, 142: w13659.