Cyanidin Analysis Service
Submit Your InquiryWhat is Cyanidin?
Cyanidin, a member of the anthocyanin family, is a natural plant pigment responsible for the red, purple, and blue hues seen in various fruits, vegetables, and flowers. Its chemical structure consists of a flavylium cation with two phenol rings and a positively charged oxygen atom. This compound is particularly abundant in foods like berries (e.g., blueberries, strawberries, blackberries), red grapes, cherries, and red cabbage, contributing not only to their vibrant coloration but also to their potential health benefits.
Research indicates that cyanidin possesses numerous beneficial properties, including antioxidant, anti-inflammatory, and anticancer activities. Additionally, it has been observed to enhance cognitive function while protecting against age-related ailments.
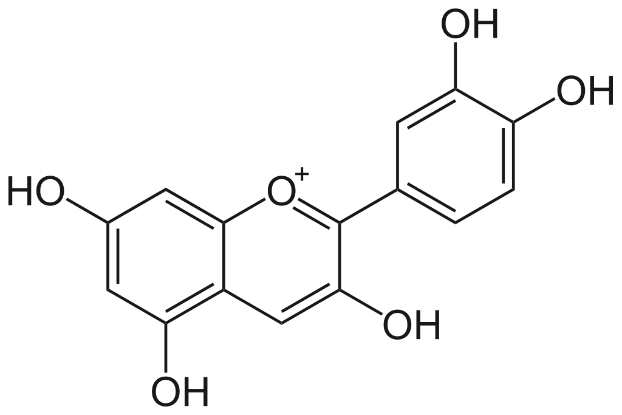 The molecular structure of cyanidin
The molecular structure of cyanidin
At Creative Proteomics, we offer a full suite of cyanidin analysis services based on mass spectrometry.
- Cyanidin Identification: We identify and characterize cyanidin metabolites, elucidating their chemical structures.
- Cyanidin Quantification: Accurate quantification of cyanidin and its metabolites in various biological matrices, facilitating pharmacokinetic studies.
- Metabolomic Pathway Analysis: We map the metabolic pathways of cyanidin, providing insights into its fate within the human body.
Our services present a vast array of advantages, foremost among them the use of cutting-edge equipment and techniques, which enable the identification of even minute quantities of cyanidin. We also customize our services to meet the unique needs of our clientele, offering everything from method development and validation to sample preparation and data analysis.
Cyanidin Analysis Platform at Creative Proteomics
Liquid Chromatography-Mass Spectrometry (LC-MS)
One of the cornerstone techniques in cyanidin metabolomics is Liquid Chromatography-Mass Spectrometry (LC-MS). This method combines the separation capabilities of liquid chromatography with the high sensitivity and specificity of mass spectrometry. At Creative Proteomics, we utilize industry-leading LC-MS instrument models, including:
- Thermo Scientific Q Exactive Series: Renowned for its exceptional resolution, accuracy, and sensitivity, the Thermo Scientific Q Exactive series allows us to detect and quantify cyanidin and its metabolites with the utmost precision.
- Waters ACQUITY UPLC-Xevo TQ-S: This high-performance system offers robust and efficient chromatographic separation coupled with the sensitivity of triple quadrupole mass spectrometry, enabling accurate quantification of cyanidin and its metabolites.
Gas Chromatography-Mass Spectrometry (GC-MS)
For the analysis of volatile cyanidin derivatives and related compounds, Gas Chromatography-Mass Spectrometry (GC-MS) is an indispensable technique. Creative Proteomics employs state-of-the-art GC-MS instrument models, including:
- Agilent 7890B GC System with 5977B Mass Selective Detector: This combination offers exceptional sensitivity and selectivity for the analysis of volatile cyanidin compounds, ensuring reliable identification and quantification.
Sample Requirements for Cyanidin Analysis
| Sample Type | Sample Matrix | Recommended Sample Volume | Storage Conditions | Special Instructions |
|---|---|---|---|---|
| Plasma/Serum | Blood | 100-200 μL per sample | -80°C or below | Collect samples in EDTA tubes to prevent coagulation. |
| Urine | Urine | 1-2 mL per sample | -80°C or below | Collect samples in sterile containers. |
| Tissues | Various (e.g., liver, etc.) | 20-50 mg per sample | Flash freeze in liquid nitrogen | Record tissue weight and source organ. |
| Cell Culture | Cell lysates | As needed | -80°C or below | Specify cell type and culture conditions. |
| Plant Extracts | Plant materials | As needed | Freeze-dried or -80°C or below | Provide details on plant species and growth conditions. |
| Food and Beverages | Various (e.g., juice, etc.) | As needed | -80°C or below or refrigerated | Include sample preparation methods and storage conditions. |
Applications of Cyanidin Analysis
Nutritional Research: Cyanidin analysis is fundamental in assessing the nutritional content of various foods and beverages, especially those rich in cyanidin, such as berries, red grapes, and red cabbage. This information is crucial for dietary recommendations and nutritional labeling.
Pharmacological Studies: Cyanidin's potent antioxidant properties make it a subject of interest in pharmacological research. Analyzing cyanidin content and its metabolites in various plant extracts or pharmaceutical formulations can assist in developing antioxidant-rich supplements or medications.
Cancer Research: The potential chemopreventive properties of cyanidin have garnered attention in cancer research. Analyzing its levels in tissues and studying its effects on cancer cells can provide valuable insights into its anti-cancer mechanisms.
Metabolism Studies: Understanding how cyanidin is metabolized in the human body is critical for pharmacokinetic studies. Analyzing its metabolism can help determine its bioavailability and the duration of its beneficial effects.
Food and Beverage Industry: Cyanidin analysis is used for quality control in the food industry. It ensures product consistency and verifies the cyanidin content in red wines, juices, jams, and fruit-based products.
Agricultural Research: Cyanidin analysis plays a pivotal role in agricultural research by aiding in the selection and breeding of plants with higher cyanidin content. This can lead to the production of crops with enhanced nutritional value and potential resistance to pests and diseases.
Pharmaceutical Development: Pharmaceutical companies may use cyanidin analysis to develop formulations that incorporate cyanidin for various therapeutic purposes.
Environmental Monitoring: Analyzing cyanidin in plants grown in different environments provides insights into the impact of environmental factors on plant metabolism and health.
Biomedical Research: Cyanidin analysis is crucial in cellular and molecular studies, helping researchers understand how cyanidin affects cellular processes and signaling pathways.
Case. Acylation of Cyanidin-3-glucoside (C3G) from Blueberry: Methods and Stability Analysis
Background
Anthocyanins, particularly Cyanidin-3-glucoside (C3G), are potent bioactive flavonoids found in fruits like blueberries. They possess attractive color properties and various health benefits due to their antioxidant and anti-inflammatory properties. However, their application as natural colorants is limited by factors such as instability and poor solubility in oils. Acylation has been proposed as a method to enhance the stability and lipotropic properties of anthocyanins, making them more suitable for food applications.
Sample
C3G was extracted from blueberry, containing 80% w/w C3G. Lauric acid was used as the acyl donor substrate, along with other reagents such as 1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide, N-hydroxybenzotriazole, and solvents including DMF and TFA.
Technical Methods
1. Chemical Acylation Procedure: C3G was combined with acyl donor substrates in DMF solvent under argon gas protection. The reaction was stirred for 48 hours at 4°C, with samples collected every 12 hours for analysis by HPLC-MS.
2. Preparative High Performance Liquid Chromatography (PHPLC) Separation and Purification: The acylation product was purified using PHPLC with an X-Bridge C18 preparative column and ACN/Water + TFA solvent system.
3. Fourier Transform Infrared (FTIR) Analysis: C3G and acylated C3G samples were analyzed using an FTIR spectrometer to characterize molecular structures.
4. Liquid Chromatography-Mass Spectrometry (LC-MS) Analysis: LC-MS was performed using an ICQ Deca ion trap mass spectrometer coupled with HPLC to analyze the acylation products.
5. Stability Analysis of Acylated C3G: The stability of acylated C3G was evaluated under different conditions, including temperature and light exposure, as well as in the presence of various additives such as vitamin C, sucrose, and glucose.
Results
Successful acylation of C3G with lauric acid was achieved, as confirmed by HPLC-MS analysis.
Purification of acylated C3G was performed using PHPLC, yielding a dry powder for further analysis.
FTIR analysis confirmed the structural changes in acylated C3G compared to unacylated C3G.
LC-MS analysis provided detailed molecular information about the acylation products.
Stability analysis demonstrated that acylated C3G showed improved stability compared to unacylated C3G under various conditions, indicating its potential as a food colorant additive
 PHPLC chromatogram of products with lauric acids as acyl donor.
PHPLC chromatogram of products with lauric acids as acyl donor.
 Total ion chromatograms of acylated C3G.
Total ion chromatograms of acylated C3G.
 Mass spectrogram of acylated C3G. Note: This was the mass spectrogram of acylated C3G, the m/z was 631.
Mass spectrogram of acylated C3G. Note: This was the mass spectrogram of acylated C3G, the m/z was 631.
Reference
- Zhao, Li-yi, et al. "Direct acylation of cyanidin-3-glucoside with lauric acid in blueberry and its stability analysis." International journal of food properties 19.1 (2016): 1-12.

